In the study, published today in the journal Cell Metabolism, they also demonstrated in animal models that it is possible to reverse this deficiency.
Chronic kidney disease affects more than 697 million people around the world. In total, it is estimated that 1.2 million people die each year due to this disease, which represents almost 5% of all annual deaths worldwide. Despite the enormous financial and personal burden that this entails, until now the biological mechanisms behind this condition were unknown, due to the structural and functional complexity of the kidney.
In a study published today in the prestigious journal Cell Metabolism, researchers from the Institute for Bioengineering of Catalonia (IBEC), led by Dr. Núria Montserrat, in collaboration with researchers from the University of Pennsylvania and the Gwangju Institute of Science and Technology, have identified the genes responsible for chronic kidney disease and have shown that it is possible to reverse this deficiency, thus opening a possible therapeutic route against kidney damage.
Using innovative techniques such as single cell RNA-sequencing, the researchers unraveled the crucial role that lipid metabolism genes play in protecting against chronic kidney failure. For this, the analyzes were carried out with animal models and human mini-kidneys. The results were validated using almost one hundred patient samples.
In healthy adults, each of our kidneys contains an average of 1.5 million nephrons, the basic functional units of the kidney that constantly filter waste products from the blood. Among the different cells that make up these filtration units, the epithelial cells of the proximal tubule of the nephron are responsible for reabsorbing water and solutes, representing 90% of the total renal mass. In almost all cases of chronic kidney disease, damage occurs in these cells, but until now, it was not known which cellular mechanisms were responsible for such dysfunction.
By sequencing the RNA of each of the thousands of cells present in the kidney, the researchers observed for the first time important differences between healthy and diseased cells in the proximal tubule. Specifically, in the kidneys of mice with chronic kidney disease, they detected that a greater proportion of the proximal tubule cells had a different molecular signature than that found in the kidneys of healthy animals.
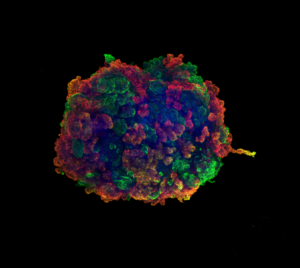
Noting these observations, the IBEC researchers went one step further and, using human mini-kidneys, demonstrated that these changes were due to a decrease in the expression of some of the genes that regulate lipid metabolism in proximal tubule cells. Furthermore, “thanks to a multidisciplinary approach, using animal models and human mini-kidneys that we generate through bioengineering at IBEC, we discovered that, by correcting this deficiency, the proximal tubule cells could regain their function in the different study models,” explains Dr. Montserrat.
Innovative analysis techniques: genetic and cell engineering
Combining single cell RNA-sequencing, and the use of bioengineering to produce mini-kidneys, they were able to identify the key cellular level differences between healthy and diseased kidneys.
Human mini-kidneys —also called kidney organoids— are created using bioengineering techniques from human stem cells, and capture some aspects of the complexity of this organ. Last April, the researchers demonstrated their effectiveness as a study model by using them to decipher in record time how SARS-CoV-2 interacts and infects the cells of these mini-kidneys, in addition to identifying a therapy aimed at reducing viral load.
In this study, single cell RNA-sequencing of the mini-kidney samples was carried out in the laboratory of Dr. Felipe Prósper at the Clínica Universidad de Navarra and CIMA Universidad de Navarra. “The kidney has more than 23 different cell types,” explains Dr. Montserrat. “A few years ago, it was necessary to examine multiple samples separately to obtain the information, which could take years of work, now this same information can be obtained in a few days”, and adds: “By RNA sequencing each kidney cell type and using multiple samples, we can identify which genes are differentially expressed in each of the cells.”
Studying these differences or, in other words, analyzing the “signature” of RNA expression in each of these cells, the researchers identified that proximal tubule dysfunction is the consequence of dysregulation of a key metabolic pathway. Specifically, in diseased mouse samples, a higher proportion of immature cells was also found. “It is known that a small proportion of the cells of the proximal tubules of the kidney exhibit a behavior similar to that of stem cells,” says Dr. Carmen Hurtado del Pozo, an IBEC researcher and co-author of the study. “When kidney damage occurs, the proximal tubule tends to respond very quickly as it can restore the parts of the nephron that are injured. However, during chronic kidney disease, these cells lose their regenerative capacity,” explains Dr. Montserrat.
The protective role of metabolism in kidney damage
This work has identified the metabolic genes that are switched off when the kidney is chronically damaged, losing the healthy “signature” in the cells of the proximal tubule of the kidney. For this signature, estrogen related receptor alpha (ESRRa) is one of the most relevant markers. “These genes are especially important for proximal tubule cells, as they need to have a very active metabolism. In fact, after heart cells, it is these cells that have the highest energy requirement in our body”, explains Dr. Montserrat. “When the expression of the genes that control lipid metabolism decreases, the kidney loses its ability to respond to damage”, adds Dr. Carmen Hurtado del Pozo.
By generating mini-kidneys, the IBEC researchers were able to simulate culture conditions in order to manipulate the levels of ESRRa in the cells of these mini-organs. When the ESRRa was switched back on, the proximal tubule appeared to regain their function. Such observations occurred in the animal models that collaborators at the University of Pennsylvania – who two years ago published for the first time the use of single-cell RNA sequencing in the kidneys of healthy mice.
This work demonstrates that the combination of emerging technologies, such as single cell RNA-sequencing and mini-organs, can be very advantageous in advancing biomedical research. This study proposes that together with ESRRa there are other nuclear receptors that may act, in the near future, as therapeutic targets to reverse chronic kidney disease.

The study was co-led by Dr. Nuria Montserrat, ICREA Research Professor and Principal Investigator of the group “Pluripotency for organ regeneration” at the Institute for Bioengineering of Catalonia (IBEC), Dr. Katalin Susztak from the University of Pennsylvania and Dr. Jihwan Park from the Gwangju Institute of Science and Technology.
Núria Montserrat is also a member of the Centro de Investigación Biomédica en Red en Bioingeniería, Biomateriales y Nanomedicina (CIBER BBN).
About the Institute for Bioengineering of Catalonia (IBEC):
The Institute for Bioengineering of Catalonia (IBEC) is a CERCA centre (Centers de Recerca de Catalunya), twice recognized as the Severo Ochoa Centre of Excellence, and a member of the Barcelona Institute of Science and Technology (BIST). IBEC conducts multidisciplinary research of excellence on the frontier between engineering and life sciences to generate knowledge, integrating fields such as nanomedicine, biophysics, biotechnology, tissue engineering and information technology applications in the field of Health. IBEC was created in 2005 by the Generalitat of Catalonia, the University of Barcelona (UB) and the Polytechnic University of Catalonia (UPC).
For more information, please contact:
Àngels López
Communication Department (IBEC)
alopez@ibecbarcelona.eu





