ABOUT
The first Open Innovation Lab research unit at IBEC will be Bioengineering in Reproductive Health @Biorephealth, led by Dr. Samuel Ojosnegros.
The main research focus of our lab is to study the development of human and mouse embryos. We combine bioengineering and optics tools to generate novel technologies that allow us to study the mechanisms controlling mammalian embryo implantation. Our proprietary solutions can be used to improve in vitro fertilization (IVF) and have been licensed by the pharma industry. The strong translational component of our research is not limited to embryology but convey into other disciplines such as diagnostics and cell therapies.
The development of the embryo E in humans (and mammals in general) requires the embryo to implantat, which proceeds with the attachment of the embryo into the walls of the mother uterus and invasion of the tissue to form the placenta. This process is crucial for natural conception and especially for in vitro fertilization (IVF) as only 30% of IVF embryos successfully implant into the mother uterus’ and develop to term. However, despite the importance of implantation in human fertility, this process is still elusive for experimentation because the uterus is rather inaccessible and difficult to be analysed
In our lab we create microscopes and bioengineered platforms to study the 3D environments that support embryonic development outside of the mother uterus. Our systems allow us to interrogation of the genetics, metabolomics and mechanics of the embryo in a high throughput manner. Using our systems, we are capable to (i) improve embryo culture conditions and (ii) diagnose embryos with improved implantation potential.
Our dual research philosophy with a basic-translational component, has granted us collaboration contracts with the pharma industry, hospitals, and venture capital to bring our technology to the clinics and to the market. Our innovative Open Lab is a multidisciplinary environment where biologists, biophysicists, clinicians, and business developers synergize to create a unique environment shaped by science and entrepreneurship.
Molecular mechanisms of embryo implantation
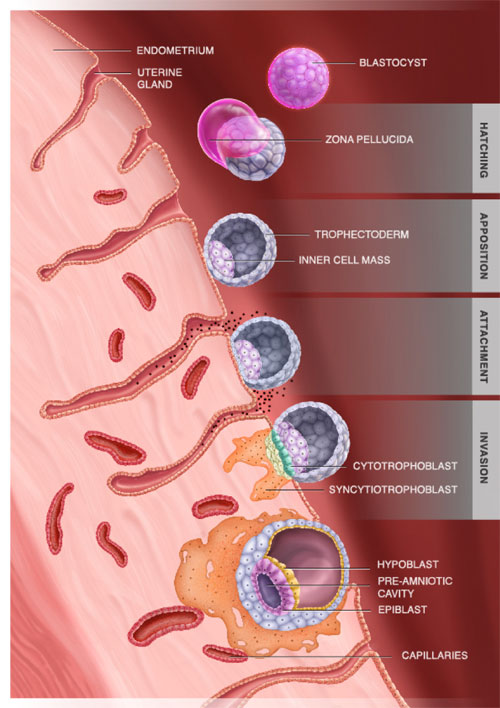
Human reproduction is surprisingly inefficient compared to other mammalian species. On average, 25–30% of natural conceptions lead to successful live births. IVF leads to a successful live birth only in about 30% of the times (averaged per embryo transferred). Around 60% of all conceptions are lost at the time of (or soon after) implantation. Implantation is not only a cornerstone of reproduction but an evolutionary novelty of mammals.
During implantation, the embryo at blastocyst stage attaches to the epithelial lining of the uterus (the endometrium). A specialized embryonic tissue, termed the trophoblast, invades the endometrium reaching out and connecting to the blood vessels. The trophoblast will give rise to the placenta and umbilical cord allowing continuous nutrient supply to the embryo and consequently a prolonged intrauterine retention of the embryo during pregnancy. However, the study of implantation is mainly inaccessible because it takes place inside the adult. To overcome this problem, we have developed proprietary culture methods and ex vivo implantation platforms which mimic the uterine microenvironment, allowing the embryo to progress towards post-implantation stages in an amenable way for optical microscopy.
Development of Human Plasma-Derived Protein Supplement to Improve Embryo Culture and Promote Embryo Implantation
Embryo culture in vitro from one cell stage to blastocyst stage in IVF clinics is carried out using human serum albumin (HSA). However non-clinical grade serums (such as FBS) have the potential to push the development of embryos further, but of course they cannot be used in the clinics. In the last years, in collaboration with GRIFOLS we have co-developed serums with batch consistency and clinical approval to culture human embryos. In addition to albumin these serums also provide growth factors, vitamins and other components that are removed during the purification process of albumin. This cocktail improves the efficiency of the development towards blastocyst stage and promote embryo ex vivo implantation.
Spectroscopy and label-free imaging
Spectroscopy refers to a family of imaging techniques which aim to extract information from the sample signal beyond the intensity profile. These techniques provide an additional dimension (i.e., spectral, oligomeric) to the image acquired.
Label-free microscopy, such as the imaging of autofluorescence signals from the cells, provide metabolic information of living tissues when analyzed using multispectral techniques.
In our lab we have specialized in spectroscopy imaging with emphasis on label-free methods. These methods are non-invasive, they do not require staining or sample manipulation whatsoever. For these reasons label-free imaging enables us to bring state-of-the-art microscopy techniques from the lab into the clinics, and study samples of biomedical relevance undisturbed at unprecedented level of detail.
Enhanced Number and Brightness a novel Imaging technique reveals protein dynamics in live cells
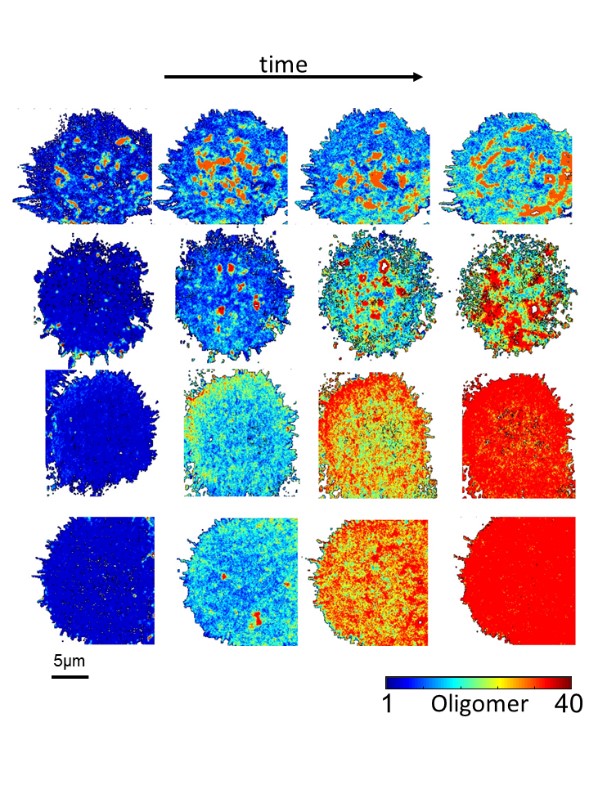
Proteins continuously interact with other proteins in the cell yet revealing these interactions in live-cell microscopy is technically challenging. Using statistical tools, we designed the Enhanced Number and Brightness method, which quantifies and maps protein aggregation in live cells in high resolution movies.
Several fluorescence microscopy methods are used to measure protein interactions in cells. However, most of these methods struggle to capture both the diversity of these interactions across the entire cellular area and their dynamics over time. To overcome these limitations, we enhanced the spatiotemporal resolution of Number and Brightness, a spectroscopy method that transforms fluorescence fluctuations into protein aggregation values.
The Enhanced Number and Brightness method implements two algorithms, the first one uses statistical tools to extract the distribution of protein oligomers (monomers, dimers, trimers and so on) in single pixels, thus revealing protein diversity across an entire cell image. The second one corrects the fluorescence intensity loss due to continuous light exposure, which enables measurements over prolonged periods of time. By combining these two algorithms, Enhanced Number and Brightness allows extended live imaging, providing dynamic maps of protein oligomerization overlaid on top of the corresponding cell image.
Figure: Enhanced Number and Brightness provides oligomerization maps (color-coded according to reference bar) during time-lapse movies. The image shows from left to right the dynamics of Eph receptor aggregation after stimulation with the ligand Ephrin. Each row depicts a cell stimulated with different types of ligands (i.e. soluble, surface-bound etc.).
STAFF
PROJECTS
European Projects
| HSMe-ImPredict · Development of non-invasive imaging methodology for improving embryo implantation prediction, via hyper-spectral metabolic profiling (2022-2024) | Marie Curie Individual Fellowship | Samuel Ojosnegros |
National projects
| HYSPLANT · Selección de embriones para fecundación in vitro: predicción del éxito de implantación mediante clasificación metabólica de embriones (2020-2023) | MINECO Retos investigación: Proyectos I+D | Samuel Ojosnegros |
| Prediction of implantation success by hyperspectral metabolic profiling of human embryos obtained by in vitro fertilization (2020-2022) | AGAUR Beatriu de Pinós 2018 | Samuel Ojosnegros |
Private sector
| Estudio del mecanismo de acción de OXO-001 (2020-2022) | Oxolife | Samuel Ojosnegros |
| Evaluación de derivados plasmáticos humanos para el cultivo de embriones (2019-2021) | IVFTECH S.L. | Samuel Ojosnegros |
| Evaluación de derivados plasmáticos humanos para el cultivo de células mesenquimales y CAR-T (2020-2022) | IVFTECH S.L. | Samuel Ojosnegros |
| Prediction of implantation success by single-cell transcriptomic profiling of human embryos obtained by in vitro fertilization (2018-2022) | Scranton Enterprises BV | Samuel Ojosnegros |
Finished projects
| HYSPLANT · Metabolic profiling of in vitro fertilization embryos using hyspectral imaging (2019-2020) | European Commission ATTRACT | Samuel Ojosnegros |
| Embryo on a chip: Smart microdevice development for high-throughput screening embryo implantation (2020) | Tecniospring 2020 | Maria Demestre Viladevall |
NEWS
Desenvolupen una tècnica pionera per saber si un òvul és bo
El grup Bioenginyeria en salut reproductiva de l’Institut de Bioenginyeria de Catalunya (IBEC) apareix als mitjans per la seva participació en el desenvolupament d’un dispositiu que ofereixi informació metabòlica de … Read more
Investigadors de l’IBEC formen part del projecte europeu ATTRACT per desenvolupar un dispositiu de diagnòstic que millori la selecció d’embrions per als procediments de fertilització in vitro
El projecte HYLIGHT, coordinat per Samuel Ojosnegros de l’IBEC, va ser seleccionat per la fase 2 de la convocatòria ATTRACT, una iniciativa de la Unió Europea per impulsar la transferència de tecnologies amb gran potencial dels laboratoris a la societat. El projecte va ser finançat amb prop de 2 milions d’euros durant 26 mesos i té com a objectiu desenvolupar un dispositiu de diagnòstic per seleccionar embrions basant-se en els seus perfils metabòlics i augmentar les taxes d’èxit de les tècniques de fertilització in vitro.
El IBEC recibe cerca de un millón de euros para estudiar las enfermedades respiratorias graves causadas por virus
El projecto REACT, liderado por el Statens Serum Institut de Dinamarca, cuenta con la participación del Instituto de Bioingeniería de Cataluña (IBEC) que, con un presupuesto cercano al millón de euros de un total de siete, generará organoides epiteliales nasales humanos específicos como plataformas de validación y modelos preclínicos para investigar la interacción huésped-patógeno .
Les teràpies regeneratives del futur van confluir al Simposi IBEC, amb experts internacionals i més de 300 inscrits
Més de tres-cents experts internacionals en el camp de la investigació en teràpies regeneratives es van citar al simposi organitzat per l’Institut de Bioenginyeria de Catalunya (IBEC) per presentar els últims avenços en miniòrgans, òrgans-en-un-xip, bioimpressió 3D i enginyeria de teixits, entre d’altres.
Els empresaris del futur visiten l’IBEC
Estudiants MBA visiten l’Institut de Bioenginyeria de Catalunya (IBEC) en una jornada organitzada per Barcelona Global.
IBEC i Oxolife inicien un estudi pioner per tractar la infertilitat
Oxolife, la startup biotecnològica focalitzada en el desenvolupament de fàrmacs per a la millora de la fertilitat femenina, i el Laboratori de Bioenginyeria en Salut Reproductiva de l’Institut de Bioenginyeria de Catalunya (IBEC), combinaran dues tecnologies pioneres per a l’estudi i tractament de la infertilitat.
Senior researcher at the Bioengineering in Reproductive Health Research Group
Application Deadline: 15/07/2020Ref: SR-SO The Bioengineering in reproductive health group at the Institute for Bioengineering of Catalonia (IBEC) is looking for a Senior Scientist to develop a project in collaboration with the industry. The project involves investigate the molecular mechanisms affected by specific chemical treatments on endometrial receptivity and on mouse and human embryos in culture. We aim to combine the expertise in microscopy of the lab with embryology tools to understand the morphological and molecular modifications that lead to an increase in endometrial receptivity. An ideal candidate will have experience in the mouse or human embryology field and will know basic tools of molecular biology. The candidate will have a great degree of independence to coordinate the project, competitive salary and funding to outsource several tests (i.e. sequencing, histology etc.). Our laboratory is a multidisciplinary environment where biologists, biophysicists, clinicians and business developers synergize to create a unique environment shaped by science and entrepreneurship. Due to the high translational component of our research, we have established collaboration contracts with the pharma industry, hospitals and venture capital to bring our technology to the clinics and the market. In our lab we use bioengineering methods to create 3D environments that support embryonic development outside of the mother uterus. Our systems are accessible to imaging tools which allow us to interrogate the genetics, metabolomics and mechanics of the embryo in a high throughput manner. Using our systems we are capable to (i) improve embryo culture conditions and (ii) diagnose embryos with improved implantation potential.
Cell culture lab technician at the Bioengineering in Reproductive Health Research Group
Application Deadline: 30/06/2020Ref: LT-SO The Bioengineering in Reproductive Health Group offers a lab technician position for an experienced cell culture technician to develop a project in collaboration with the industry. The project involves testing the effects of different cell culture supplements on several cell types (e.g. Mesenchymal Stromal Cells, CAR T Cells, etc.) Our laboratory is a multidisciplinary environment where biologists, biophysicists, clinicians and business developers synergize to create a unique environment shaped by science and entrepreneurship. Due to the high translational component of our research, we have established collaboration contracts with the pharma industry, hospitals and venture capital to bring our technology to the clinics and the market. In our lab, we develop high-tech projects in the fields of embryology, cell biology and imaging. Our strategy involves the development of new products for the pharmaceutical industry. The current position seeks a motivated cell culture technician with experience in diverse cell culture systems.
L’IBEC signa un acord per 1.5 milions d’euros amb Scranton Enterprises
L’Institut de Bioenginyeria de Catalunya (IBEC) ha signat un acord per una ronda d’inversió inicial d’un milió i mig d’euros amb Scranton Entreprises B.V. -una associació d’inversió amb seu als Països Baixos-, amb la finalitat de desenvolupar tecnologia que predigui la probabilitat d’implantació d’embrions obtinguts mitjançant la fecundació in vitro. Aquest acord és el primer que s’estableix en el marc de la nova iniciativa de l’institut, l’Open Innovation Lab, que ofereix a empreses i inversors una plataforma per a portar de manera més àgil la recerca d’avantguarda als pacients i al mercat.
Desxifrant el llenguatge de les cèl·lules
Conèixer el mecanisme de comunicació intercel·lular que regula el posicionament de cèl·lules obre camí cap al desenvolupament de teràpies dirigides en medicina regenerativa Conèixer el llenguatge de les cèl·lules per poder-les redirigir cap a on sigui necessari. Aquesta és una de les possibilitats obertes pels investigadors del Centre de Medicina Regenerativa de Barcelona (CMR[B]), liderats pel Dr. Samuel Ojosnegros, que han aconseguit descriure en el seu últim estudi el mecanisme de comunicació intercel·lular implicat en el posicionament de les cèl·lules. El treball, publicat per la prestigiosa revista Proceedings of the National Academy of Sciences (PNAS) , compta amb la col·laboració dels grups d’Elena Martínez (IBEC) i Melike Lakadamyali (ICFO), entre d’altres. La fructífera col·laboració també ha donat lloc a la publicació del treball de la Verónica Hortigüela, ex estudiant de doctorat al grup de l’Elena, que ha dissenyat un patró nanomètric de proteïnes que permet controlar aquest mecanisme de comunicació.
JOBS
Embryology Laboratory Technician at the Bioengineering for Reproductive Health Research Group
Ref: PD-SO/Deadline: 04/04/2024
Open postdoc position on embryology at the Bioengineering in Reproductive Health Research Group
Ref: PD_SO/ Deadline: 12/02/2024
Research Assistant at the Bioengineering for Reproductive Health Research Group
Ref: RA_SO/Deadline: 20/11/2023
Postdoc on microscopy at the Bioengineering in Reproductive Health Research Group
Ref: PD_SO//Deadline: 1/11/2023
Postdoctoral researcher at the Bioengineering in Reproductive Health Open Lab (Ref: PD_SO)
Ref: PD_SO // Deadline: 19/01/2023
Embryologist at the Bioengineering in Reproductive Health Research Group (Ref: LT_SO)
Ref: LT_SO // Deadline: 19/01/2023
Biochemist at the Bioengineering in Reproductive Health Research Group
Reference: LT-SO / Until Oct 15th, 2022
PUBLICATIONS
S Ojosnegros, A Seriola, AL Godeau, A Veiga (2021) Embryo implantation in the laboratory: an update on current techniques. Human Reproduction Update, Vol.00, No.0, pp. 1–30.
Martin Plöschner, Denitza Denkova, Simone De Camillis, Minakshi Das, Lindsay M. Parker, Xianlin Zheng, Yiqing Lu, Samuel Ojosnegros, and James A. Piper (2020) Simultaneous super-linear excitation-emission and emission depletion allows imaging of upconversion nanoparticles with higher sub-diffraction resolution. Optics Express 28 (16), 24308-24326.
EQUIPMENT
- Micromanipulation-microinjection station
- Embryo biopsy laser
- Embryo culture laboratory
- Genome editing
- In vitro fertilization
- ICSI
- Cell culture laboratory
- 3D cell culture
- Advanced live imaging: photoconversion, 3D imaging, light scattering, spectroscopy
- Image analysis workstation
COLLABORATIONS
- Prof. Anna Veiga – Barcelona Stem Cell Bank (IDIBELL) and Dexeus Mujer, Barcelona
- Dr. Montserrat Boada/ Dr. Pere Barri – Dexeus Mujer, Barcelona
- Dr. Ayelet Lesman – Tel Aviv University (TAU), Israel
- Dr. Elena Martínez – IBEC
- Dr. Francesco Cutrale, University of Southern California (USC), USA
- Dr. Manuel Irimia – CRG, Barcelona
- Dr. Javier Ramón – IBEC
ENTREPRENEURSHIP
· Jorge Fuentes,
Business Strategy, A_Ventures, Barcelona, Spain
 |  | ||
 |  |
Follow us on Twitter: @Biorephealth

