About
We aim at understanding how physical forces and molecular control modules cooperate to drive biological function.
We develop new technologies to map and perturb the main physical properties that determine how cells and tissues grow, move, invade and remodel.
By combining this physical information with systematic molecular perturbations and computational models we explore the principles that govern the interplay between chemical and physical cues in living tissues.
We study how these principles are regulated in physiology and development, and how they are derailed in cancer and aging.
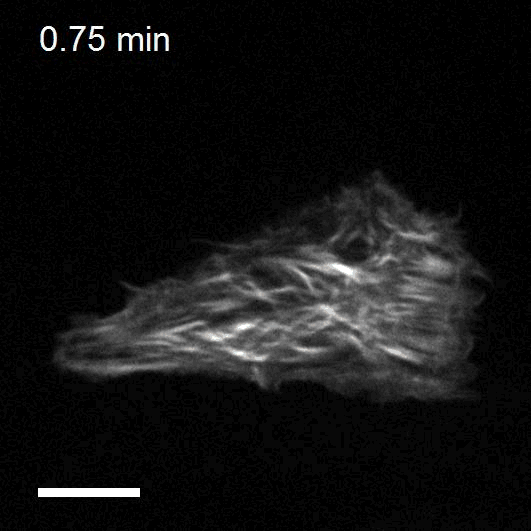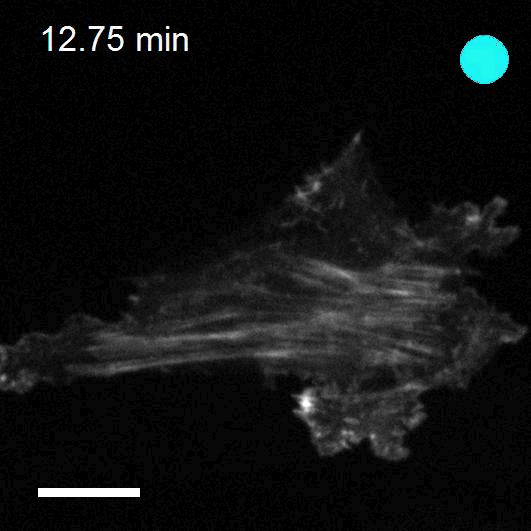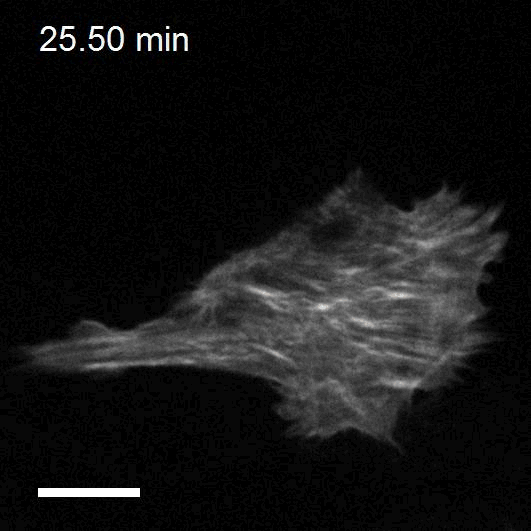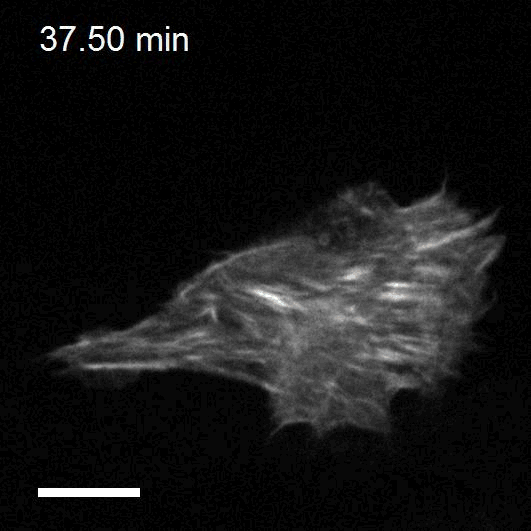
Making cellular forces visible
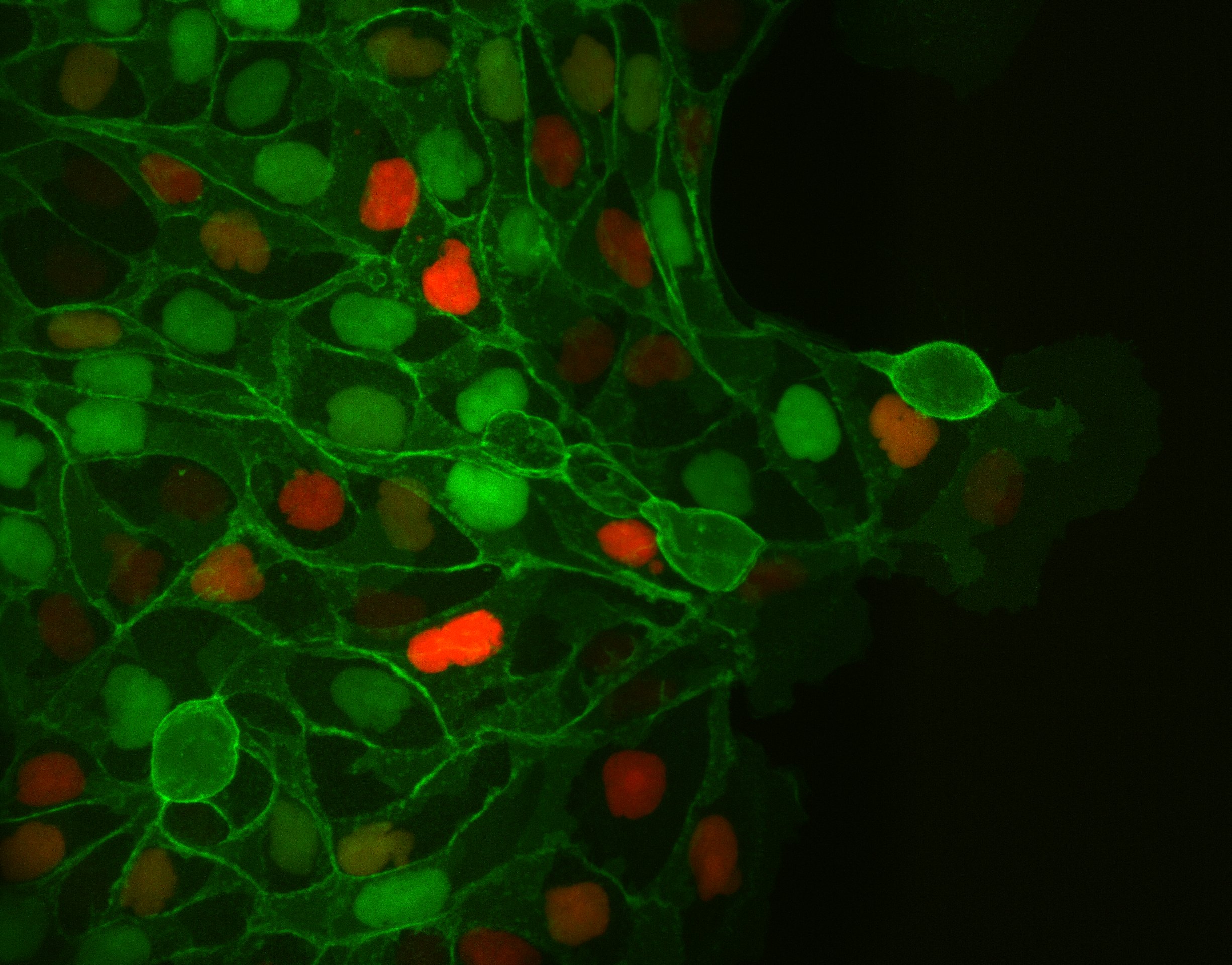
To study cell and tissue dynamics we develop new technologies to measure physical forces at the cell-cell and cell-matrix interface. By combining these technologies with computational analysis of cell shape and velocity we obtain a full experimental characterization of epithelial dynamics during tissue growth, wound healing and cancer cell invasion.
Tumour invasion by stromal forces
Cancer cell invasion and metastasis remain the leading cause of death in patients with cancer. Both processes are the result of a complex interaction between tumor cells and their microenvironment. One of our main lines of research is to study how tumours exploit the functions of non-cancer cells in their microenvironment to invade and metastasize. We focus on the interaction between epithelial cancer cells and Cancer Associated Fibroblasts (CAFs), the most abundant cell type in the tumour stroma.
Optogenetics to control cell mechanics
The recent development of optogenetic technologies offers promising possibilities to control signalling pathways with high spatiotemporal resolution. By expressing genetically encoded light-sensitive proteins, optogenetic technology enables the reversible perturbation of intracellular biochemistry with subcellular resolution. We have developed optogenetic tools based on controlling the activity of endogenous RhoA to upregulate or downregulate cell contractility and to control cell shape and mechanotransduction.
Collective durotaxis: a mechanism for cellular guidance by mechanical cues
Directed cell migration is one of the earliest observations in cell biology, dating back to the late XIX century. Also known as taxis, directed cell migration has been commonly associated with chemotaxis, i.e. the ability of a broad variety of cell types to migrate following gradients of chemical factors. We recently demonstrated a new mode of collective cell guidance by mechanical cues, called collective durotaxis. This new migration mode emerges only in cell collectives and, strikingly, does not require isolated cells to exhibit gradient sensing.
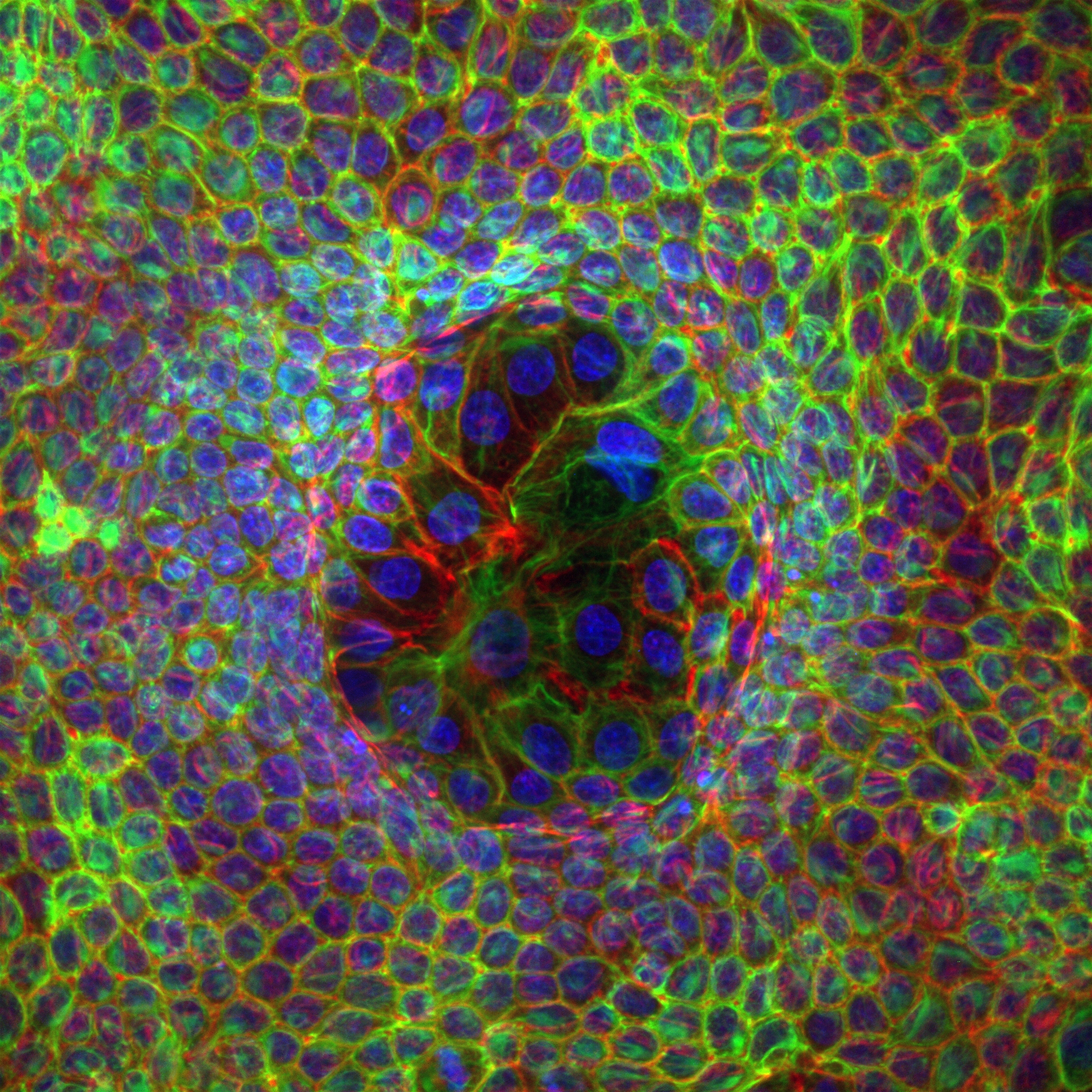
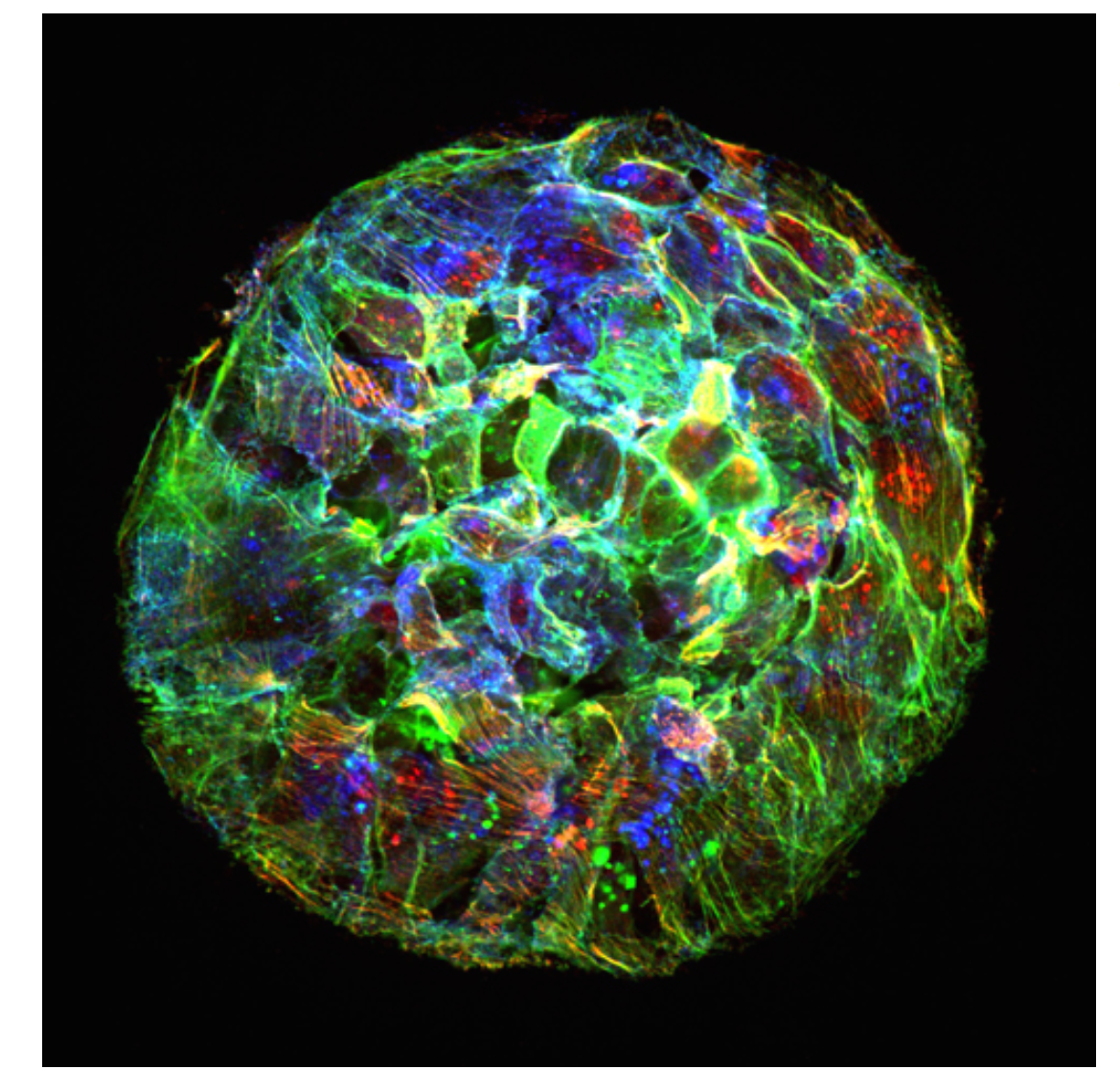
Organoid mechanobiology
Organoids are large multicellular structures that self-organize in vitro and maintain a similar organization and functionality than the organ from which they are derived. Organoids from many organs have now been obtained from embryonic stem cells, induced pluripotent stem cells and organ progenitors. We use intestinal and kidney organoids to study how epithelia adopt three-dimensional shapes that closely resemble their structure in vivo. We also use organoids grown from primary tumors to understand how epithelial structure and function are lost with disease progression.
Engineering epithelial shape and mechanics from the bottom up
We develop new approaches to engineer epithelia in 3D. Using these approaches, we study the principles that govern the emergence of tissue shape from the bottom up. We recently found that epithelial sheets can stretch up to four times their initial area without breaking, and that they are able to recover their initial size in a fully reversible way when unstretched. Surprisingly, some cells in the tissue barely stretch, while others become ‘superstretched’, increasing their area more than ten times. We call this phenomenon ‘active superelasticity’.
Staff
Xavier Trepat
Projects
| NATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| mGRADIENTMecanobiología de la migración colectiva durante la haptotaxis y la durotaxis: aplicación a los organoides intestinales (2019-2022) | MICIU Generación Conocimiento: Proyectos I+D | Xavier Trepat |
| DYNAGELHidrogeles biocompatibles con rigidez dinámicamente ajustable para estudiar la mecanobiología de células y tejidos (2019-2022) | MICIU Retos investigación: Proyectos I+D | Raimon Sunyer |
| INTERNATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| EpiFold Engineering epithelial shape and mechanics: from synthetic morphogenesis to biohybrid devices (2021-2025) | European Commission, ERC-AdG | Xavier Trepat |
| The role of intermediate filaments in stress resistance in 3D epithelial structures (2021-2023) | Deutsche Forschungsgemeinschaft (DFG), Walter Benjamin-Programme | Tom Golde |
| Mechano·Control Mechanical control of biological function (2017-2022) | European Commission, FET Proactive | Xavier Trepat |
| Control of cell collective flows and tissue folding by means of surface patterns (2021-2022) | Human Frontier Science Program, HFSP Beca postdoctoral | Pau Guillamat |
| PRIVATELY-FUNDED PROJECTS | FINANCER | PI |
|---|---|---|
| Mech4Cancer · Enabling technologies to map nuclear mechanosensing: from organoids to tumors (2020-2023) | Obra Social La Caixa: Health Research Call | Xavier Trepat |
| T cell exclusion during cancer immune evasion and immunotherapy failure: cell types, transcriptional programs and biomechanics (2020-2023) | Fundació La Marató de TV3 | Xavier Trepat |
| Joint Programme Healthy Ageing | Obra Social La Caixa | Xavier Trepat |
| Understanding and measuring mechanical tumor properties to improve cancer diagnosis, treatment, and survival: Application to liquid biopsies (2017-2022) | Obra Social La Caixa | Xavier Trepat |
| FINISHED PROJECTS | FINANCER | PI |
|---|---|---|
| OPTOLEADER Optogenetic control of leader cell mechanobiology during collective cell migration (2019-2021) | European Commission, MARIE CURIE – IF | Leone Rossetti |
| MECHANOIDS Probing and controlling the three-dimensional organoid mechanobiology (2019-2021) | European Commission, MARIE CURIE – IF | Manuel Gómez |
| TensionControl Multiscale regulation of epithelial tension (2015-2020) | European Commission, ERC – CoG | Xavier Trepat |
| El mecanoma de la adhesión epitelial: mecanismos de detección, resistencia y transmisión de fuerzas intercelulares | MINECO, I+D-Investigación fundamental no orientada | Xavier Trepat |
| MICROGRADIENTPAGE Micro Gradient Polyacrylamide Gels for High Throughput Electrophoresis Analysis | European Commission, ERC-PoC | Xavier Trepat |
| GENESFORCEMOTION Physical Forces Driving Collective Cell Migration: from Genes to Mechanism | European Commission, ERC-StG | Xavier Trepat |
| CAMVAS Coordination and migration of cells during 3D Vasculogenesis (2014-2017) | European Commission, MARIE CURIE – IOF | Xavier Trepat |
| DUROTAXIS Mecanobiología de la durotaxis: de las células aisladas a los tejidos | MINECO, Proyectos I+D Excelencia | Xavier Trepat |
Publications
Equipment
- Soft Lithography
- Micro/Nano fabrication
- Cell stretching
- Live Confocal Microcopy
- Magnetic Tweezers
- Magnetic Twisting Cytometry
- Monolayer stress microscopy
- Traction microscopy
Collaborations
- Julien Colombelli / Eduard Batlle
Institute for Research in Biomedicine (IRB) Barcelona - Marino Arroyo
Universitat Politècnica de Catalunya, Barcelona - Guillaume Charras / Roberto Mayor
University College London, UK - Erik Sahai
Cancer Research, UK - Benoit Ladoux
Université Paris 7, France - Jim Butler & Jeff Fredberg
Harvard University, Boston - Danijela Vignjevic
Institut Curie, Paris - Jonel Trebicka
Department of Internal Medicine I, University Hospital Frankfurt
News
Xavier Trepat guanya el Premi Fundació Lilly de Recerca Biomèdica 2024
Xavier Trepat, professor ICREA de l’IBEC ha estat guardonat en la categoria de recerca preclínica. Amb aquest premi, la Fundació Lilly reconeix el treball de Trepat, distingit per l’aplicació de tècniques i conceptes de la física a les ciències de la salut.
La interdisciplinarietat de l’IBEC protagonitza la secció d’opinió de Nature Physics
Teresa Sanchis i Xavier Trepat, membres de l’IBEC, han publicat sengles articles d’opinió a la secció “World View” de la prestigiosa revista científica Nature Physics. Ambdós articles ressalten el compromís de l’IBEC amb l’excel·lència científica, el foment de la diversitat i la creació d’un entorn propici per a la col·laboració, així com el rellevant paper de l’IBEC a escala internacional en recerca multidisciplinària.
La Bioenginyeria es converteix en art al Museu Antoni Tàpies
Una instal·lació ubicada a l’exposició “A=A, B=B” a la Fundació Antoni Tàpies permetrà accedir en directe als laboratoris de l’IBEC. Una finestra des d’on els visitants tindran l’oportunitat de veure com els investigadors desenvolupen els seus projectes de recerca, com si es tractés d’una autèntica performance d’innovació científica.
Evitar que els teixits responguin al seu enduriment pot ser clau per frenar l’avanç dels tumors de mama
Un estudi liderat per l’Institut de Bioenginyeria de Catalunya demostra que la laminina, una proteïna present en els teixits mamaris, evita els efectes de l’enduriment, protegint les cèl·lules contra el creixement tumoral. El mecanisme s’ha demostrat in vitro, però es creu que també podria funcionar in vivo a partir de l’observat en mostres de pacients.
El rellotge intern de les nostres cèl·lules es veu afectat per les forces mecàniques
Investigadors de l’IBEC demostren com les forces físiques desregulen el rellotge circadiari de les cèl·lules, el mecanisme que controla els canvis fisiològics cada 24h. La troballa pot ajudar a entendre … Read more
Daniel Navajas: 30 anys dedicats a la mecanobiologia
El passat 5 de maig es va celebrar el simposi Before Mechanobiology had a name (abans que la mecanobiologia tingués un nom), amb motiu de la jubilació del Professor Daniel … Read more
Les cèl·lules canceroses es traslladen a entorns rígids com si fossin gotes
Investigadors de l’Institut de Bioenginyeria de Catalunya (IBEC) i la Universitat de Barcelona (UB) han creat un nou marc teòric i experimental que explica la durotaxi col·lectiva, un mecanisme de … Read more
Mecanotransducció als mitjans
Investigadors de l’IBEC liderats per Pere Roca-Cusachs i Xavier Trepat apareixen als mitjans per un estudi que obre portes per a noves investigacions de teràpies i diagnòstics contra el càncer.
EMBL-IBEC Conference als mitjans
L’EMBL-IBEC Conference, una conferència d’una durada de tres dies organitzada per l’Institut de Bioenginyeria de Catalunya (IBEC) i el Laboratori Europeu de Biologia Molecular (EMBL), apareix al diari ARA on experts internacionals han discutit sobre com reproduir malalties humanes en sistemes vius generats al laboratori.
L’investigador Xavier Trepat guanya el premi “Constantes y Vitales” per les seves contribucions pioneres al camp de la mecanobiologia
El Professor de Recerca ICREA a l’Institut de Bioenginyeria de Catalunya (IBEC), Xavier Trepat, ha estat guardonat amb el Premi “Constantes y Vitales” al Talent en Investigació Biomèdica, atorgat pel grup Atresmedia, per les seves aportacions al camp conegut com a mecanobiologia, aquell que estudia l’efecte de les forces físiques sobre els sistemes vius i la seva implicació en patologies com ara el càncer.
Jobs
Postdoc at the Integrative Cell and Tissue Dynamics Research Group
Ref: XT-PD/Deadline: 18/12/2023
Research assistant at the Integrative Cell and Tissue Dynamics Research Group
Ref: RA_XT/Deadline: 15/06/2023
Senior researcher at the Integrative Cell and Tissue Dynamics Research Group (SRR_XT)
Ref SRR_XT // Deadline : 16/01/2023
Laboratory Technician at the Integrative Cell and Tissue Dynamics Research Group (LT_XT)
Ref: LT-XT // Deadline: January 16th
Postdoc at the Integrative Cell and Tissue Dynamics Research Group (Ref: PD3-XT)
Ref: PD3-XT / Deadline: 20/12/2022
Research Assistant at the Integrative Cell and Tissue Dynamics Group
Ref: RA2-XT // Deadline: 09/12/2022
Postdoc for the synthetic morphogenesis of epithelial tissues at the Integrative Cell and Tissue Dynamics Research Group
Postodoctoral / Deadline: November 23th 2022
Postdoc to the mechanics of cellular monolayers under optogenetic control at the Integrative Cell and Tissue Dynamics Research Group
Postdoctoral / Deadline: 23th November 2022
Research Assistant at the Integrative Cell and Tissue Dynamics Group
Research Assistant / Deadline: November 22nd 2022


 ibecbarcelona.eu
ibecbarcelona.eu