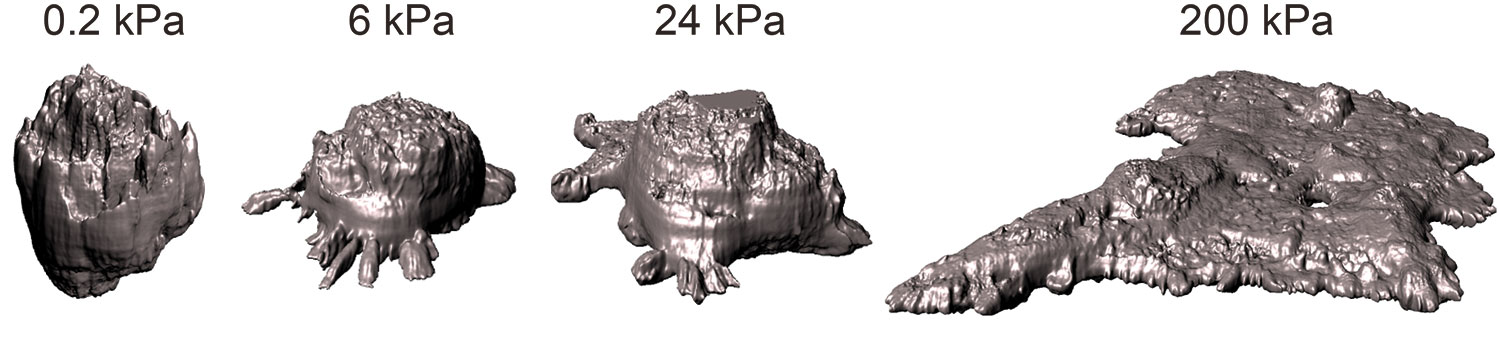
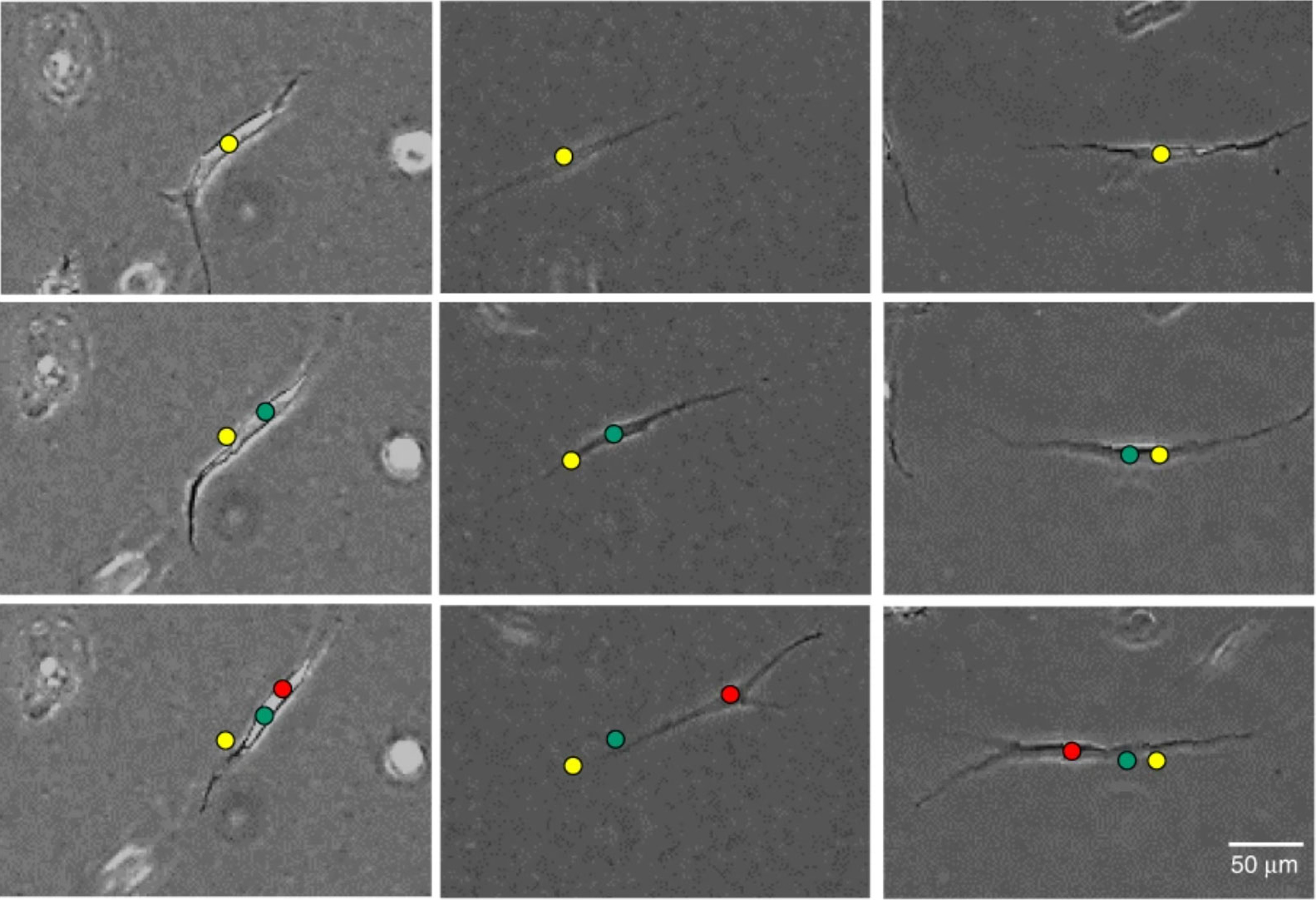
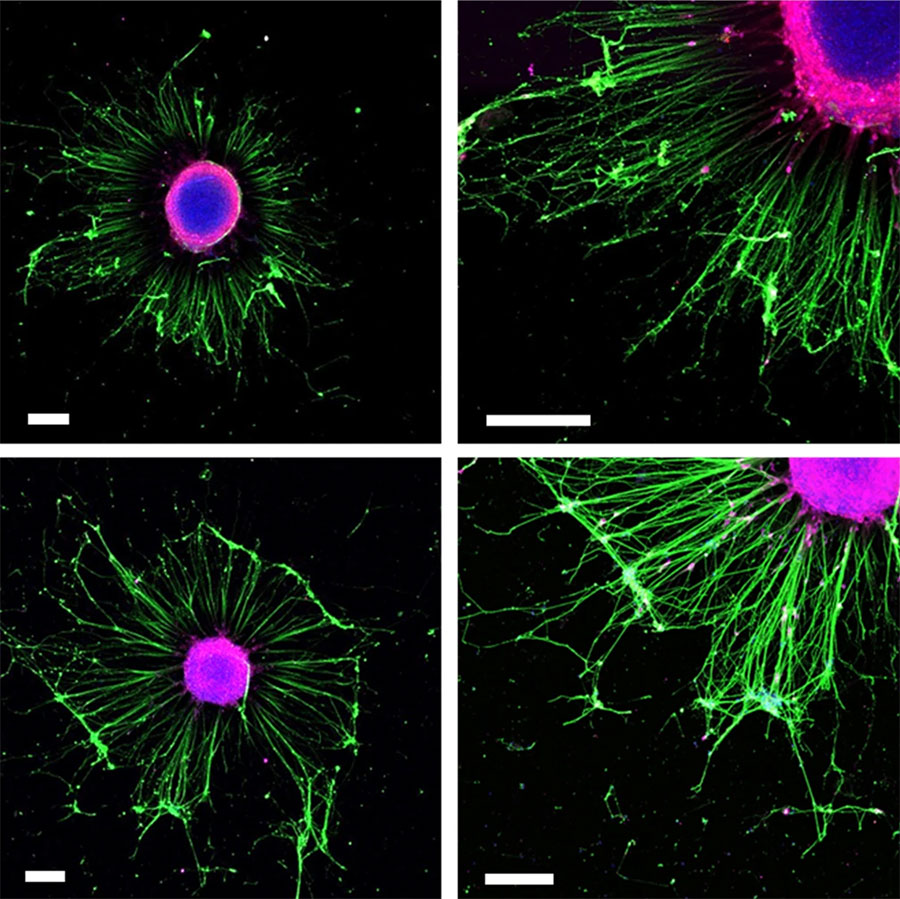
Researchers identify new biomarkers of the most frequent lung cancer
Researchers from the IBEC and the University of Barcelona (UB) have identified new biomarkers for non-small cell lung cancer, the most common lung cancer. The results, published in the journal Modern … Read more