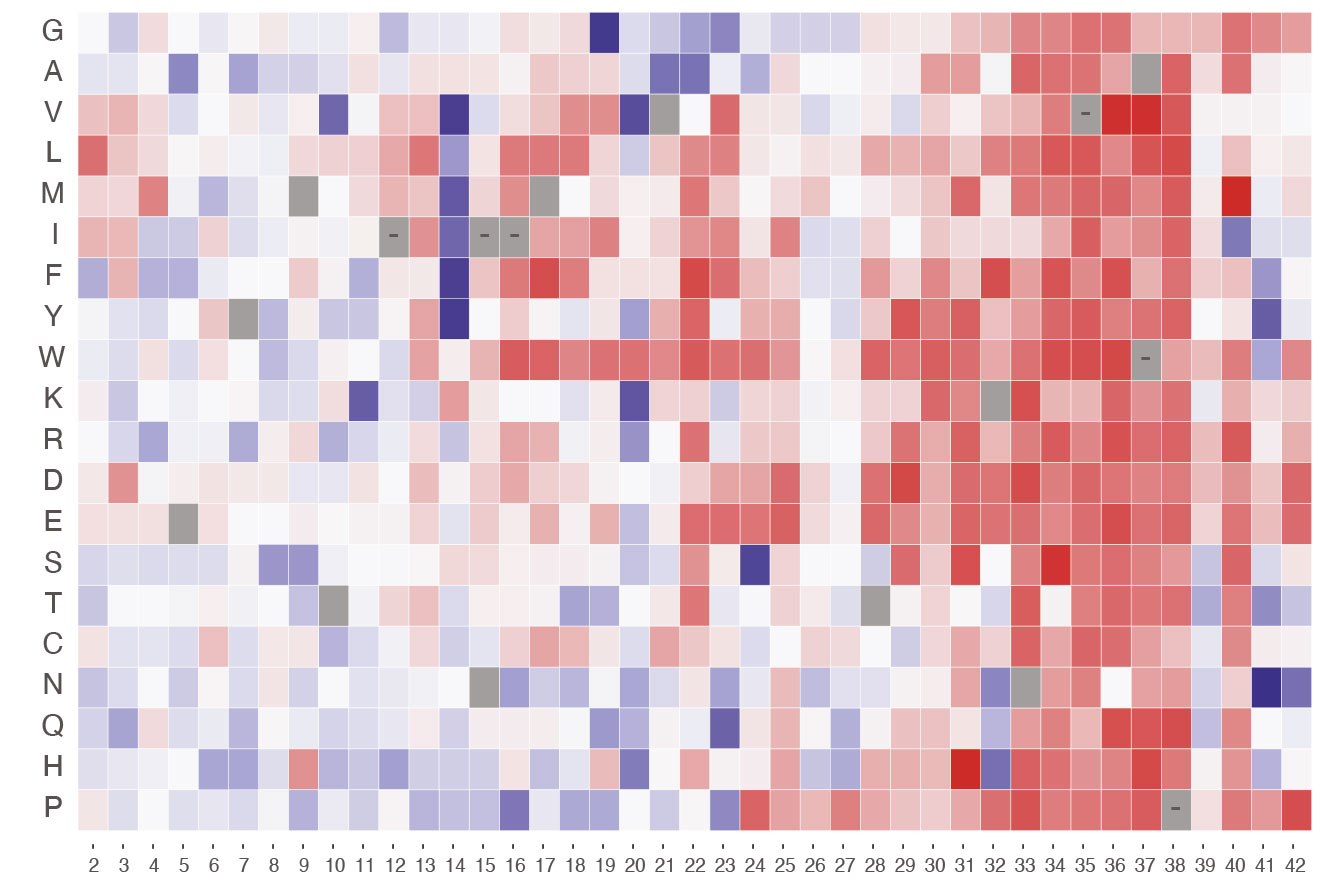
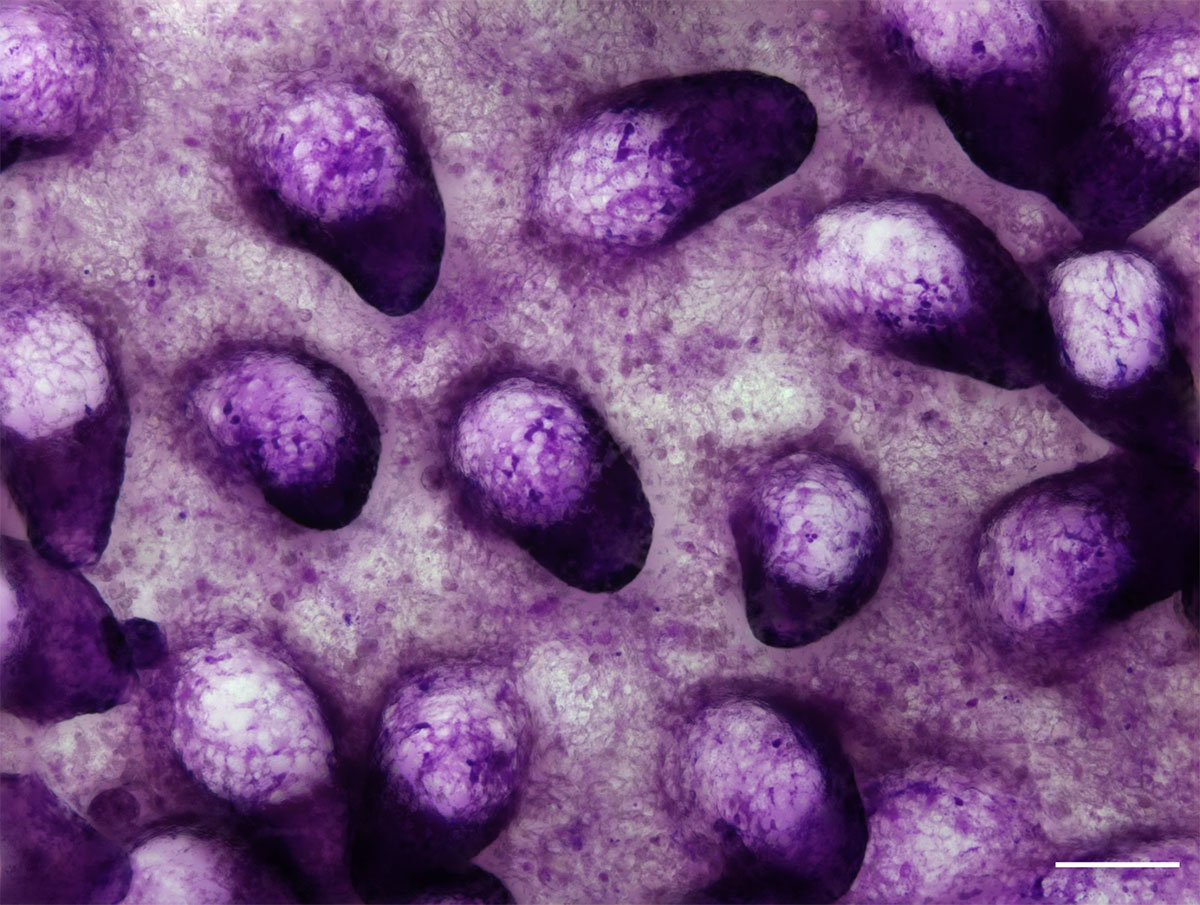
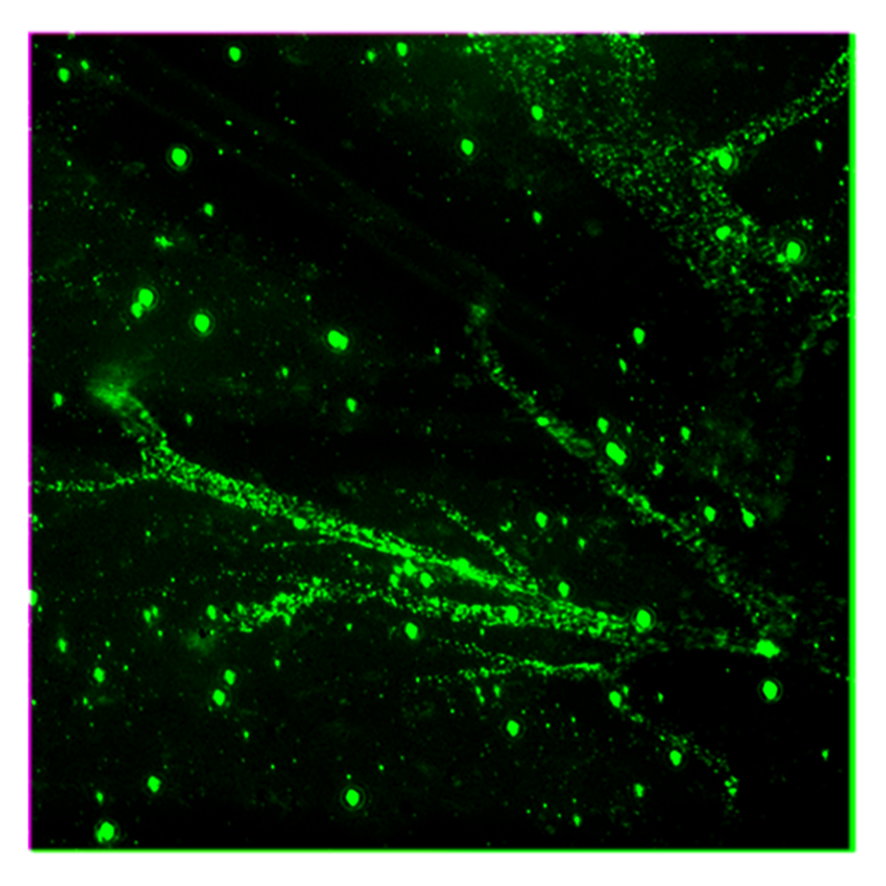
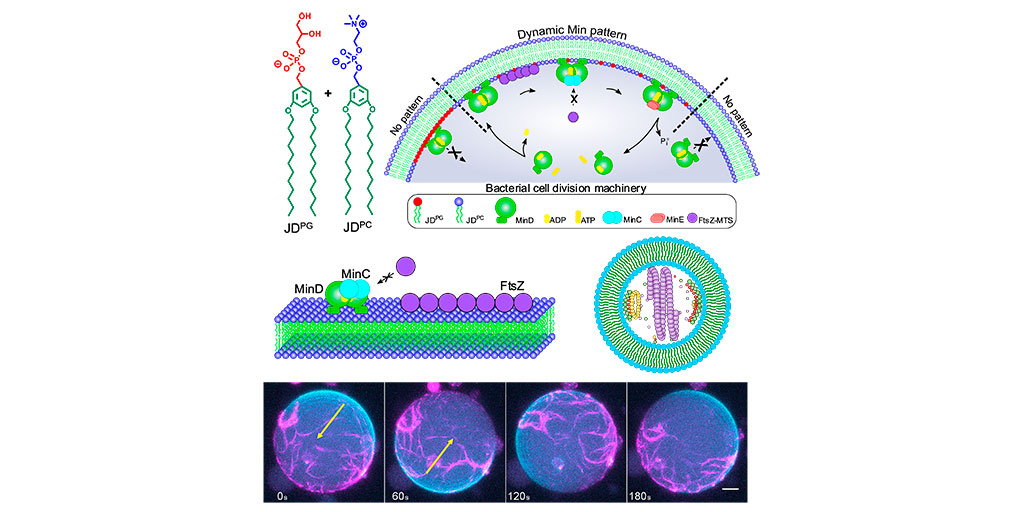
Nanoscale discovery reveals how phosphorylation affects electron transport in mitochondria
Researchers at the Institute for Bioengineering of Catalonia (IBEC) and the Biomedical Research Networking Center in Bioengineering, Biomaterials, and Nanomedicine (CIBER-BBN) have achieved a nanoscale view of the electron transfer … Read more