Group: Bacterial infections: antimicrobial therapies
Group leader: Eduard Torrents (etorrents@ibecbarcelona.eu)
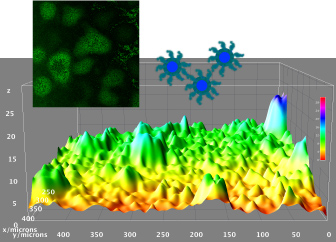
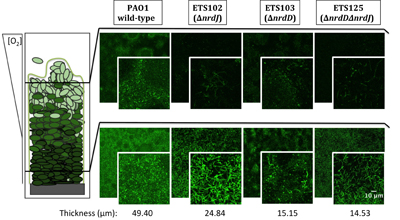
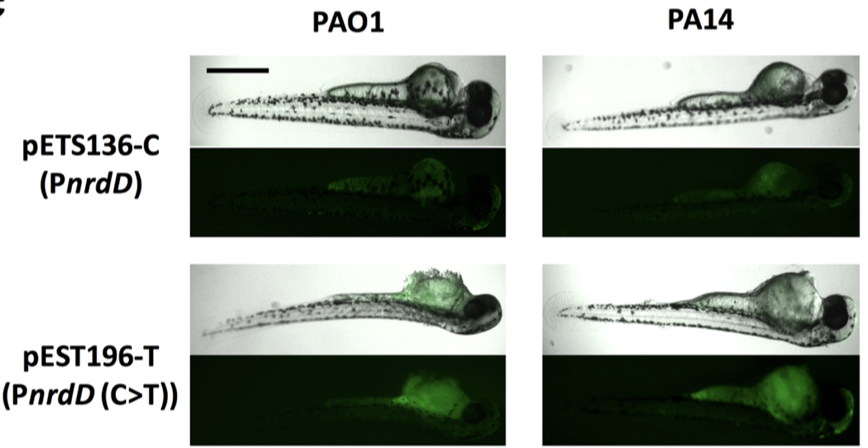
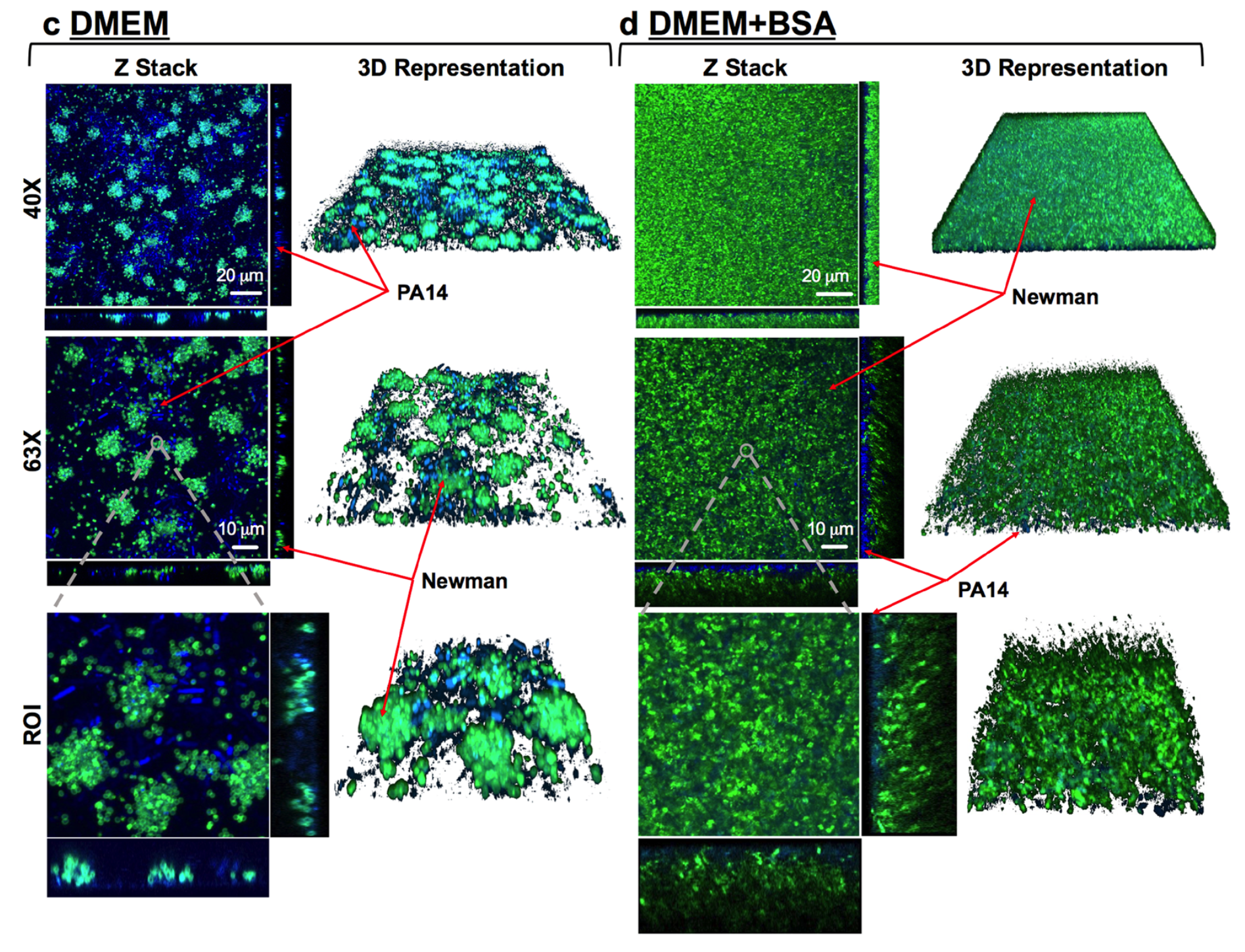
Infectious diseases are the leading cause of death worldwide. Disease-causing bacteria that resist antibiotic treatment are now widespread in every part of the world and have reached “alarming levels” in many areas, as stated by the World Health Organization. “The problem is so serious that it threatens the achievements of modern medicine,” entering to A post-antibiotic era in which common infections and minor injuries can kill. Nowadays, bacterial biofilm-based infections have emerged as a significant public health concern.
We aim to explore and characterize new antimicrobial molecules that specifically inhibit new antibacterial targets. Also, we are exploring new antimicrobial strategies based on nanoparticles to deliver new and pre-existing antimicrobials with the capacity to evade the immune system and able to penetrate formed bacterial biofilms. The group is focused on the study of mixed biofilms and study the interaction between two different bacteria. Fundamental studies in microbial physiology, new vaccines, molecular microbiology and microfluidics applied to the microbiology diagnostic are also performed in our laboratory. See further details and publications at our group web page: www.torrentslab.eu.