Research news
Researchers discover how cellular membranes change curvature depending on BAR proteins
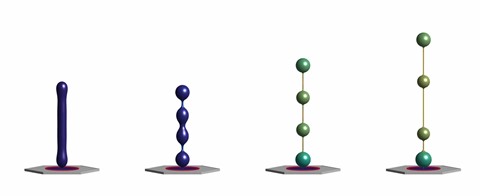
A team of researchers at IBEC and UPC, led by Pere Roca-Cusachs and Marino Arroyo, study how BAR proteins, a family of molecules that bind curved cellular membranes, reshape these membranes. Scientists report in the journal Nature Communications, through both experiments and modelling, the dynamics of these membrane reshaping processes that occur both in normal cells or disease scenarios.
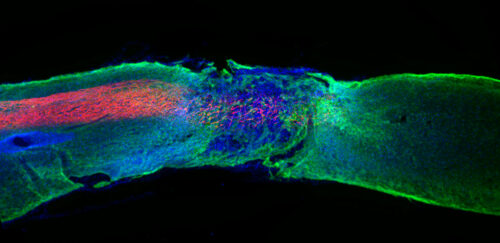
‘Dancing molecules’ repair severe spinal cord injuries in paralyzed animals
Researchers from the University of Northwestern, in the United States, and the Institute of Bioengineering of Catalonia (IBEC), develop a new injectable therapy that uses synthetic nanofibers to repair spinal cord lesions. Nanofibers, which contain ‘Dancing molecules’ effectively stimulate cellular receptors by promoting spinal cord regeneration. After a single injection, paralyzed animals treated with this new therapy were able to walk again in just four weeks. The article has been published in Science.
Artificial smell to control food quality
Researchers from IBEC, in collaboration with the University of Cordoba, recently published a study where they develop protocols that optimize the use of a technique capable of analysing, at the molecular level, substances present in the aroma of food, managing to differentiate samples of ham from Iberian pigs fed with acorn or feed. This new approach, which uses artificial intelligence to analyse the data, will simplify the analysis of aromas, and can be very useful to determine the traceability and quality of food, and fight against fraud.
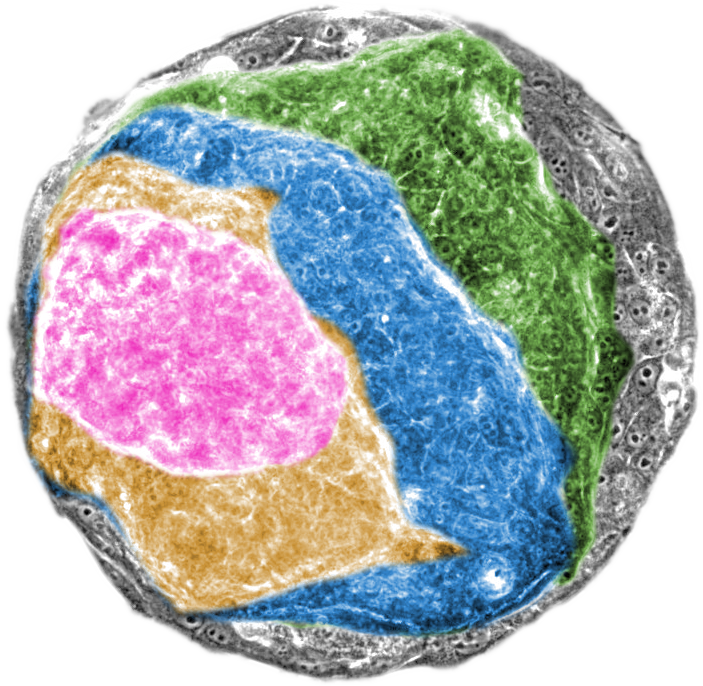
RAS gene and tissue physics act in tandem to drive cancer processes
Researchers from IBEC, the UCL (UK), UPC (Spain) and the TU/e (Netherlands) show that activation of the RAS oncogene (a mutation occurring in approximately 30% of human cancers), induces tumour features in an epithelium via coordinated physical changes of all cells forming the affected tissue. Published in the scientific journal Science Advances, the new work suggests that future cancer treatments should target both the biological processes underlying the disease and the evolving mechanical structure of the affected tissue.
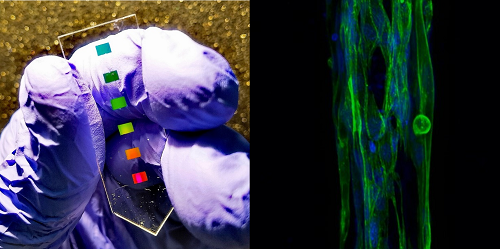
New biosensor detects inflammatory marker in muscle with high sensitivity
In a recent publication in the journal Nanophotonics, IBEC researchers present a new biosensor for the direct and sensitive detection of the protein interleukin-6 in muscle, an indicator of inflammation and potential disease, proving the high performance of the device on bioengineered 3D skeletal muscles. This new approach may result in a promising tool for measuring the efficacy of drug candidates for diseases where inflammation is present such as muscular dystrophy.
Personalised medicine to beat pediatric leukemia
IBEC researchers have used a state-of-the-art analysis technique to evaluate new drug combinations to successfully treat acute lymphoblastic leukemia. This study brings personalised medicine closer to the clinic for this cancer type.