About
We are chemists, physicists, mathematicians, engineers, biologists who work alongside to design bionic units that mimic specific biological functions and/or introduce operations that do not exist in Nature. We apply a constructionist approach where we mimic biological complexity in the form of design principles to produce functional units from simple building blocks and their interactions. We called such an approach: Molecular Bionics.
We are engaged in several activities involving the synthesis and characterisation of novel hierarchal materials whose properties are the result of the holistic combination of its components:
Molecular Engineering
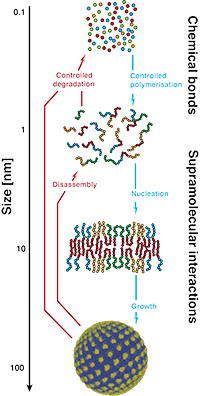
We combine synthetic and supramolecular chemistry to tune inter/intramolecular interactions and self-assembly processes to form dynamic soft materials whose molecular, supramolecular and mesoscale structures are tuned and fit for the final application (pictured right: molecular engineering of nanoscopic structures starting from molecule passing to polymers and finally to supra molecular structures).
Physical Biology
Our materials are designed to interact with living systems and thus its biological activity is studied in high detail. We have developed and established new methodologies to study living systems and how synthetic materials interact with them combining holistically physical and life sciences (Physical Biology).
Synthetic Biology
Both know-hows are applied to study biological organisation and complexity creating synthetic surrogates that act as models, as well as to engineer novel sophisticated ways to interact with living organisms.
Somanautics
In analogy to medical bionics, where engineering and physical science converge to the design of replacement and/or enhancement of malfunctioning body parts, we take inspiration from viruses, trafficking vesicles and exosomes to apply molecular engineering to create nanoscopic carriers that can navigate the human body (Somanautics) with the final aim to improve drug delivery or create new diagnostic tools.
Visit our external website to find out more.

Staff
Giuseppe Battaglia
Projects
| INTERNATIONAL GRANTS | FINANCER | PI |
|---|---|---|
| CheSSTag · Chemotactic Super-Selective Targeting of Gliomas (2020-2023) | European Comission / ERC-CoG | G. Battaglia |
| NATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| BrainPePN · Nanomedicinas de precisión que penetran el cerebro (2021-2024) | MICIU / Retos investigación: Proyectos I+D | G. Battaglia |
| FUNDRAISING PROJECTS | FINANCER | PI |
|---|---|---|
| A por la COVID-19 (2021-2022) | IBEC / Faster Future 2020 | G. Battaglia |
Publications
Check for more detailed information on the outputs of the Group at IBEC CRIS portal.
Publications list:
Equipment
- State-of-the-art facilities for cell culture including 5 class A cell cabinets: one dedicated for LPS and RNAse free cell culture and one dedicated for infected tissues
- Fluorescence Activated Cell Sorting (FACS)
- Confocal microscope to perform live cell 4D imaging
- Thermocycler
- Real-time PCR
- Automated Western Blot
- Gel Permeation Chromatography
- High-Performance Liquid Chromatography
- Ultra Performance Liquid Chromatography equipped with fluorescence, UV/Vis and Infrared and light scattering detectors
- Dynamic light scattering unit
- Nanoparticle tracking analysis
- UV and Fluorescence spectroscopy
- Automated liquid handling units
- Nanoparticle production units
Collaborations
- Xavier Salvatella
IRB Barcelona - Francesca Peiro
Physics-University of Barcelona - Kostas Kostarellos
Life Science- University of Manchester/ICN2 - Giorgio Volpe
Chemistry-UCL - Simona Parrinello
Cancer Institute -UCL - Finn Werner
Structural Biology -UCL - Nick Lane
Evolutionary Biology -UCL - Darren Hargraves
Pediatric Neuro-Oncology -UCL - Timothy McHugh
Clinical Microbiology =UCL - Sebastian Brander
Neurology -UCL - Joan Abbott
Physiology -King’s College London - Molly Stevens
Bioengineering -Imperial College London - Stefano Angioletti-Uberti
Materials Science -Imperial College London - Ricardo Sapienza
Physics -Imperial College London - Daan Frenkel
Chemisty-University of Cambridge - Charlotte Williams
Chemistry -University of Oxford - Francesco Gervasio
Pharmacology -University of Geneve/UCL, UK - Francesco Stellacci
Bionegineering -EPFL Switzerland - Tambet Tessalu
Cancer Biology -University of Tartu (Estonia)/ Sanford Burnham Prebys Medical Discovery Institute - Darrel Irvine
Bioengineering -MIT - Xiaohe Tian
Life Sciences University of Anhui - Yupeng Tian
Chemistry University of Anhui - Lei Luo
Pharmacy -Southwest University, China - Kai Luo
HuaXi hospital Sichuan University - Darren Hargrave
Great Ormond Street Hospital, UCLH London - Sebastian Brander
Queen Square National Centre for Neurology, UCLH London
News

BIST calls for reforms to turn scientific discoveries into European industry
The BIST Forum 2026 brought together scientists and representatives from academic, economic and social institutions to discuss how frontier research can help build Europe’s industries of the future. The event also announced the recipients of the BIST To the Mothers of Science and Ignite programmes.

Amayra Hernández-Vega receives a grant from the Pasqual Maragall Foundation to advance research on the early stages of Alzheimer’s
IBEC researcher Amayra Hernández-Vega has been awarded a grant from the Pasqual Maragall Researchers Programme to lead a project focused on studying the earliest stages of Alzheimer’s disease. The study will combine in vitro and human models of familial Alzheimer’s disease to identify age-related factors and potential inhibitors that could slow the early progression of the disease. The project was presented at an informative session on sleep and brain health, which was attended by around 500 people.

The IBEC and West China Hospital hold their third joint conference on precision medicine in Barcelona
Barcelona hosted the third IBEC-WCH Precision Medicine Conference this week, an event that further strengthened the strategic alliance between the Institute for Bioengineering of Catalonia (IBEC) and the West China Hospital of Sichuan University (WCHSU). The Chinese delegation visited Spain from 26 to 29 November, taking part in a busy schedule of scientific and institutional activities and exchanges between the two centres.

IBEC research takes third place in the 2025 Vanguardia de la Ciencia Awards
A study led by IBEC has won the 3rd Vanguardia de la Ciencia 2025 Prize. The research, led by Samuel Sánchez Ordoñez and with Meritxell Serra-Casablanca as lead author, proposes an innovative bladder cancer therapy based on nanorobots loaded with radiopharmaceuticals. These nanorobots are capable of moving around the bladder using urine as a source of energy.

LA VANGUARDIA: ¿Dianas artificiales para hacer llegar medicamentos al cerebro?
Daniel Gonzalez-Carter, investigador del Instituto de Bioingeniería de Catalunya (IBEC), lidera el proyecto brain-TAG.
La Razón: Un estudio hispano-chino abre la vía para tratar el alzhéimer con nanotecnología
Diseñan nanopartículas que eliminan los tóxicos que dañan al cerebro y restauran habilidades mentales que parecían perdidas.

IBEC, one of the centres selected by the Pasqual Maragall Foundation for its research grants
Amaya Hernández-Vega, a senior researcher at IBEC, will lead one of the two projects selected in the 2024 call for proposals from the Pasqual Maragall Research Programme, which is dedicated to funding research into Alzheimer’s disease and other neurodegenerative conditions. IBEC also celebrates the participation of Jordi Duran, an associate researcher from the Molecular and Cellular Neurobiotechnology group, who will collaborate on the second selected project.
ARA: Una injecció reverteix símptomesde l’Alzheimer en ratolins
L’administració de nanopartícules permet netejar toxines del cervell
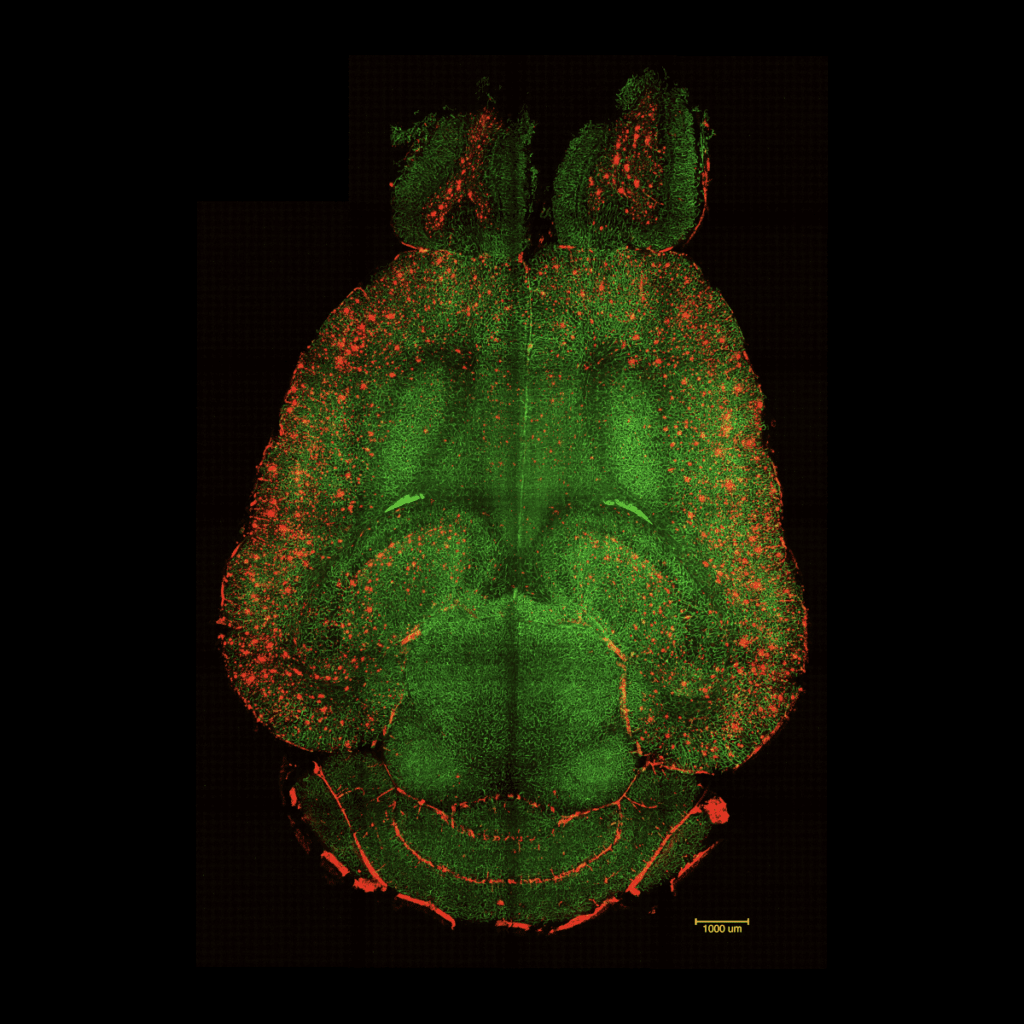
EL PERIÓDICO: Científicos de BCN y China revierten el alzhéimer en ratones
Diseñan una novedosa técnica centrada en restaurar la salud vascular con inyecciones de nanopartículas. «En una hora se han empezado a ver resultados».

Scientists reverse Alzheimer’s in mice using nanoparticles
An international team co-led by the Institute for Bioengineering of Catalonia (IBEC) and the West China Hospital Sichuan University (WCHSU) was able to reverse Alzheimer’s in mice after only 3 injections with nanoparticles. The innovative approach to treating the disease focuses on restoring the normal function of the vasculature, rather than acting on neurons or other brain cells, as has usually been done until now. This achievement, published in the Signal Transduction and Targeted Therapy Journal, is a promising step towards an effective treatment for Alzheimer’s disease.
Jobs
Postdoctoral Researcher at the Molecular Bionics Research Group
Ref: PDR-GB // Deadline: 28/02/2026
Laboratory Technician at the Molecular Bionics Research Group
Ref: LT-AH // Deadline: 24/02/2026
Predoctoral researcher at the Molecular Bionics Research Group (Project TauiST)
Ref: FPI_GB // Deadline: 29/10/2025
Postdoctoral Researcher at the Molecular Bionics Research Group
Ref: PR_GB // Deadline: 11/08/2024
Postdoctoral Researcher at the Molecular Bionics Research Group (PD_GB)
Ref: PD_GB // Deadline: 24/05/2024
Senior Laboratory Technician at the Molecular Bionics Research Group (SLT_GB)
Ref: SLT_GB // Deadline: 24/12/2023
Research Assistant at the Molecular Bionics Research Group (RA_GB)
Ref: RA_GB // Deadline 12/12/2023


 ibecbarcelona.eu
ibecbarcelona.eu