To carry out the study, published today in the prestigious journal Cell, the experts have used mini-kidneys created from human stem cells generated at IBEC by Nuria Montserrat’s team. These organoids, which have been obtained applying bioengineering techniques, reproduce the complexity of the real organ and allowed the researchers to decipher how SARS-Co-V2 interacts and infects human kidney cells. Moreover, they have validated a therapy able to reduce substantially viral load of COVID-19.
“The use of human organoids allows us to test in a very agile way the treatments that are already being used for other diseases or that are close to being validated. When time is short, these 3D structures dramatically reduce the time we would spend trying a new drug on humans” states Núria Montserrat.
The role of the ACE2 receptor in SARS-Co-V2 infection
Recent publications have shown that to infect a cell, coronaviruses use a protein, called S, that binds to a receptor on human cells called ACE2 (angiotensin-converting enzyme 2). Considering that this binding has been identified as a gateway for the virus to enter the body, avoiding it could constitute a possible therapeutic target.
Following this strategy, the researchers explain that they have focused on understanding the role of the receptor ‘ACE2 in these human organoids because they mimic, in a few millimeters almost all the characteristics of a real organ. The study provides new insights on key aspects of SARS-Co-V2 and its interactions at a cellular level, and also how the virus can infect the blood vessels and kidneys.
In addition to the lung, the ACE2 receptor is expressed in multiple tissues, including the heart, blood vessels, intestine, and kidneys, which would explain the multi-organ dysfunction seen in patients. The fact that this receptor is strongly expressed in the kidneys and that SARS-Co-V2 can be found in urine, is what has led the researchers to use these human kidney organoids as a testing model, whose creation Montserrat is an international benchmark.
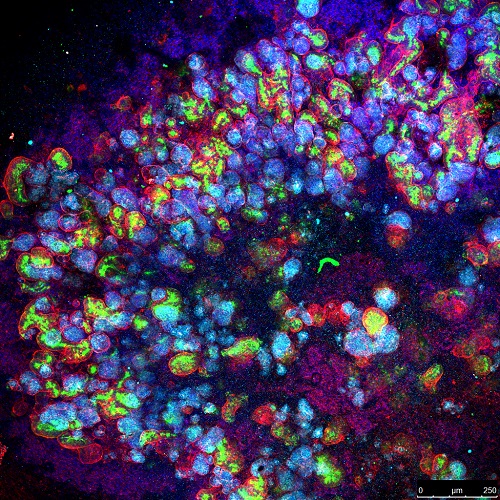
Identifying an infection-inhibiting drug
First, the researchers proved that the kidney organoids contained different groups of cells that expressed ACE2 in a similar way to that seen in the native tissue, and then proceeded to infect it with SARS-CoV-2. Once they obtained these infected mini-kidneys, they applied different therapies, and, as a result of the study, they concluded that hrsACE2 (human recombinant soluble ACE2), a drug that has already passed phase 1 clinical trials (in healthy volunteers) and phase 2 (in patients with acute respiratory distress syndrome), it significantly inhibits SARS-CoV-2 infections and reduces their viral load.
“These findings identify a promising treatment capable of stopping the infection of the new coronavirus in early stages, which, as of March 30, has affected more than 750,000 people and more than 36,000 lives have been lost due to Covid-19 worldwide,” concludes Núria Montserrat.
Findings like this show that bioengineering techniques are essential for the medicine of the future. Until now, they have allowed the creation of organoids and organs on a chip, among others. Likewise, molecular engineering, which is closely linked to bioengineering, has been proving for years its ability to validate the efficacy of experimental treatments and reduce animal experimentation. Now these tools are made available to society once again to try to find solutions to the coronavirus crisis.
Núria Montserrat leads the Pluripotency for organ regeneration group at IBEC and is a member of the Center for Network Biomedical Research in Bioengineering, Biomaterials and Nanomedicine (CIBER BBN). Apart from Núria, Elena Garreta, Patricia Prado and Carmen Hurtado, from her research group at IBEC, have also contributed to this work.
Reference article: Monteil et al, Inhibition of SARS-CoV-2 infections in engineered human
tissues using clinical-grade soluble human ACE2, Cell 2020, DOI: 10.1016/j.cell.2020.04.004
More information:
Àngels López · alopez@ibecbarcelona.eu





